Reagents

About us | Contact us | Ordering | Home


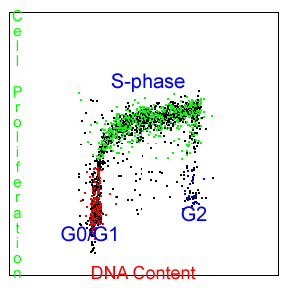
One of the most easily measured features of cell proliferation is the changing amount of cellular DNA content associated with each of the cell cycle phases. Cellular DNA content may be labeled with any of a variety of DNA binding fluorochromes and the subsequent fluorescence measured to determine the relative DNA content and cell cycle position of the cells being studied. This measurement is called cell cycle analysis. Cell cycle analysis is only a snapshot of what percentage cells are in each compartment of the cell cycle. An experimenter can not determine what percentage of the cells are actual going through the cell cycle and synthesizing more DNA at a given time point with this assay (proliferating). In a DNA content assay, the cells could be confluent (not turning over) and stuck in S-phase or G2 phase. This type of measurement is inherently somewhat imprecise, though, with the cutoffs between the G0/G1-, S- and G2/M-phases of the cell cycle not clearly delineated on the flow cytometry data. For the past 25 years we have been selling our DNA cell cycle analysis software, MultiCycle AV, to perform this analysis. This is not a cell proliferation measurement.
It is often desirable to have a means of unequivocally identifying actively proliferating cells and the rate at which these cells are proliferating. The acceptable scientific method for doing this is to specifically label newly replicated DNA for a short period of time and then measure the newly replicated DNA.
 .
.
Though there are several approaches to accomplishing this, the methods of choice involve some how labeling the nucleotide building blocks that are incorporated during DNA replication. An ingenious method for allowing this has been developed, involving the use of 5-bromo-2-deoxyuridine (BrdU), a thymidine analog. As a thymidine analog, BrdU is preferentially incorporated into newly replicated DNA(instead of thymidine)and can then be subsequently fluorescently labeled, unequivocally identifying cells containing such DNA. The growth of cells in the presence of 5-bromo-2-deoxyuridine (BrdU) has become an accepted method for monitoring DNA replication.
The use of BrdU for assaying DNA replication has replaced methods utilizing radioisotope-labeled thymidine in both research and clinical laboratories. The incorporation of BrdU into cellular DNA is most commonly detected using anti-BrdU antibodies. This methodology requires that the cellular DNA be slightly denatured to separate the duplex strands in order for the BrdU epitope to become accessible and reactive to the antibody. This denaturation process usually involves a heat treatment (>90C), acid (2-4N HCl) treatment, enzymatic (DNAse digestion) or UV light exposure (SBIP). Each method has its proponents and detractors and no denaturation method has been proven to be clearly superior to any of the others. For this reason, PFS offers two kits presently. One uses the UV light exposure (SBIP) method (ABSOLUTE-S) and the other uses the acid denaturation method (EZ-BrdU). Proponents of the enzymatic treatment method maintain that the acid treatment results in the loss or denaturation of many cellular proteins. However, using the “low acid” method of the EZ-BrdU kit, we have not seen this limitation and have found it well suited for simultaneous staining of many intracellular proteins. The EZ-BrdU method is definitely less labor intensive and gives better DNA cell cycle results than either the enzymatic or SBIP method but does not simultaneously measure apoptosis and cell proliferation as the SBIP method does. We leave it up to the individual investigator to decide which method is best for his particular experimental goals. We only strive to provide the least confusing and least cumbersome protocols for each of the assays, EZ-BrdU & ABSOLUTE-S(SBIP) Kit configurations.
PFS has two cell proliferation labeling kits: EZ-BrdU and ABSOLUTE-S. Both kits can be used for flow cytometry or fluorescent image cytometry. The normal configuration of the assay is to measure cell proliferation with a green dye (FITC) and the cell cycle with a red dye (Propidium Iodide). Our cell proliferation reagent kits have all necessary reagents including the Propidium Iodide solution that the customer needs to perform the application and also contain positive and negative controls to eliminate any guesswork in the staining procedure (these features are in contrast to products from other manufacturers).
In addition, PFS offers multiple configurations of the assay kit for investigators wishing to use the green fluorescence parameter of the flow cytometer for another measurement. For example, if the flow cytometer has a second laser (Red HeNe) in addition to the standard blue argon, the EZ-BrdU kit can be ordered with an Alexa-647 conjugation of the anti-BrdU antibody. We also feature a red conjugation which can be measured in the phycoerytherin channel of a flow cytometer. Also offered are most of the Alexa dye conjugations from Invitrogen, including the Alexa 488. These are useful in image cytometry for their superior resistance to fading under a light source.